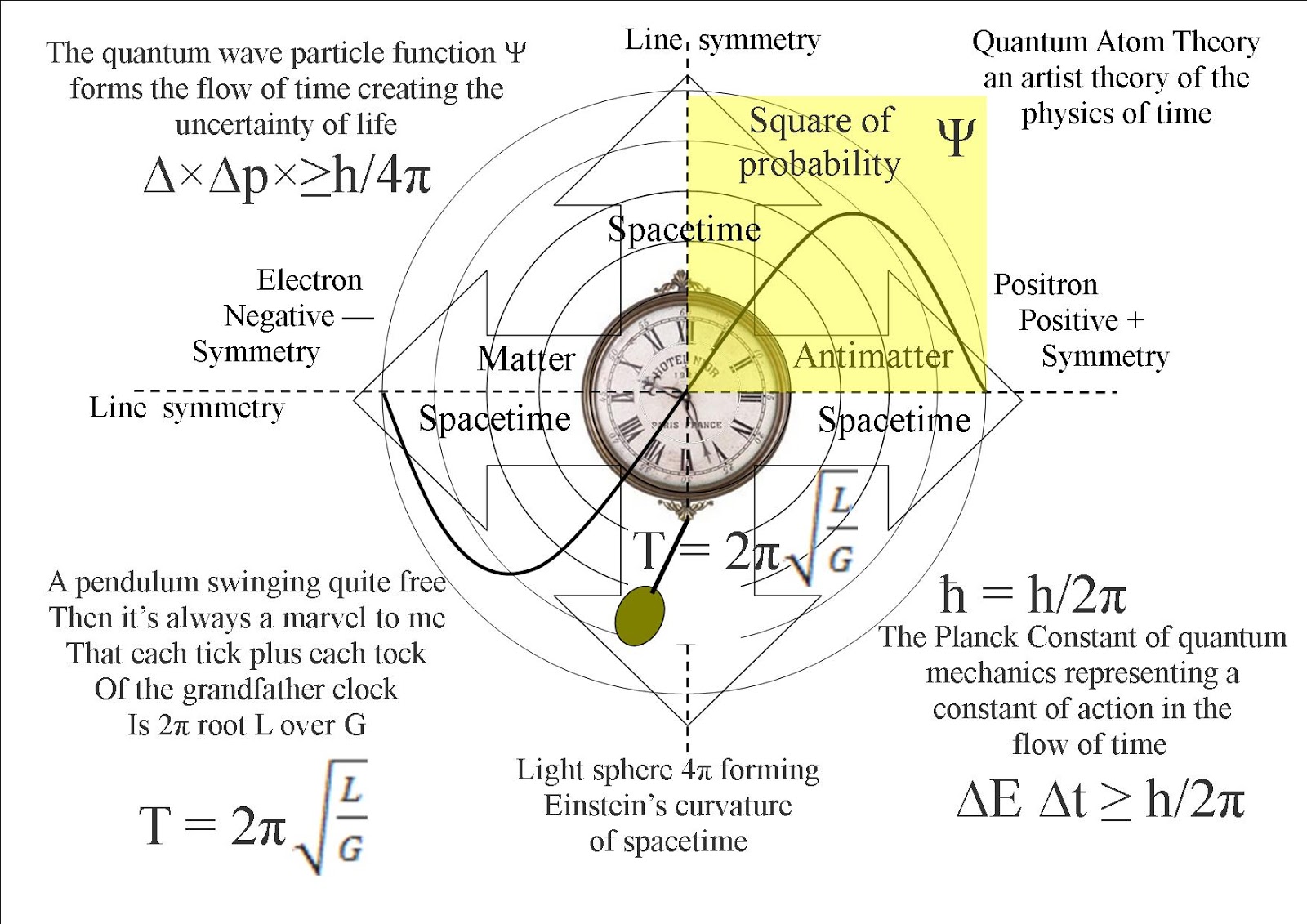
In the published 1927 paper, Heisenberg originally concluded that the uncertainty principle was Δ pΔ q ≈ h using the full Planck constant. Explore the Heisenberg uncertainty principle by calculating uncertainty in position given the uncertainty in momentum for Bohr model of hydrogen. Certain pairs of measurements such as (a) where a particle is and (b) where it is going (its position and momentum) cannot be precisely pinned down. The Heisenberg uncertainty principle states that there is a limit to how precisely certain pairs of physical properties of a particle can be known simultaneously.  
It relates to measurements of sub-atomic particles. Clausius-Clapeyron equation phase diagram for a pure substance phase. Introduced first in 1927 by German physicist Werner Heisenberg, the uncertainty principle states that the more precisely the position of some particle is determined, the less precisely its momentum can be predicted from initial conditions, and vice versa. The uncertainty principle, also known as Heisenbergs uncertainty principle, is a fundamental concept in quantum mechanics. Heisenberg's uncertainty principle is one of the most important results of twentieth century physics. Heisenbergs uncertainty principle, Schrodinger wave equation (time independent). Chad Orzel navigates this complex concept of quantum physics. Why not Because everything in the universe behaves like both a particle and a wave at the same time. Such paired-variables are known as complementary variables or canonically conjugate variables. The Heisenberg Uncertainty Principle states that you can never simultaneously know the exact position and the exact speed of an object. More formally, the uncertainty principle is any of a variety of mathematical inequalities asserting a fundamental limit to the product of the accuracy of certain related pairs of measurements on a quantum system, such as position, x, and momentum, p. In other words, the more accurately one property is measured, the less accurately the other property can be known. The virtue of Schrodingers version (5) is that it accunts for this correlation. It states that there is a limit to the precision with which certain pairs of physical properties, such as position and momentum, can be simultaneously known. The uncertainty principle, also known as Heisenberg's uncertainty principle, is a fundamental concept in quantum mechanics. According to Hunds rule, as electrons are added to a set of orbitals of. 2: The 2p 2 p sublevel, for the elements boron (Z 5) ( Z 5), carbon (Z 6) ( Z 6), nitrogen (Z 7) ( Z 7), and oxygen (Z 8) ( Z 8). Uncertainty principle of Heisenberg, 1927. The figure below shows how a set of three p p orbitals is filled with one, two, three, and four electrons. Canonical commutation rule for position q and momentum p variables of a particle, 1927.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
